Licensed Stem Cell Therapy in Mexico
People like having options. It is natural. Part of the result is the journey; as such, we tend to search for different possibilites when choosing which therapy best fits us because, fundamentally, we want to heal, not just treat the symptoms of our ailments.

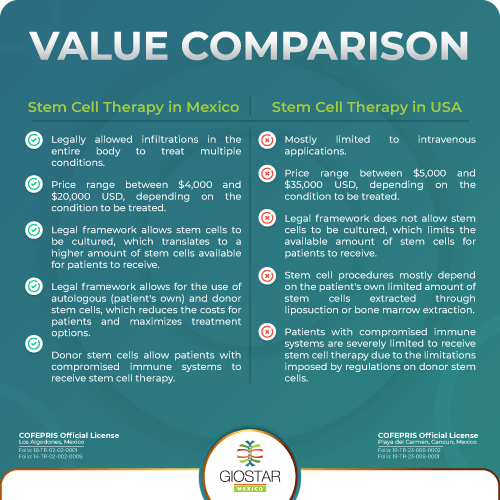
Stem cell therapy in Mexico is thoroughly regulated by the Mexican FDA-equivalent: COFEPRIS. Due to advanced legislation, Mexico enjoys a progressive legal framework that allows applications otherwise unavailable in countries such as the US or Canada. For example: local direct applications at hands, shoulders, knees, are prohibited in some states of the US or, where allowed, limited to autologous stem cells (patient’s own, which require a previous minor liposuction surgery and need up to 1 month for cultivation of stem cells in laboratory); lumbar and cervical applications, as well as Intrathecal (IT) applications, which have demonstrated better results for patients with spinal cord injury, ALS and MS, among other conditions, are banned in the US and Canada.

It is common to receive inquiries from patients looking for stem cell therapy for ALS, Arthritis or COPD, among other conditions that we generally treat. Although stem cell therapy is a field within Regenerative Medicine that has been authorized (albeit limited to IV applications and certain conditions) for commercial offering in the US since bone marrow transplants were generalized in the 20th century, it is still sometimes considered “alternative” or “new” by the general public. Arguably, Mexico is a convenient healthcare service tourist destination for US citizens, due to geographic proximity, affordable costs, rapid acces and availability of stem cell therapy applications due to progressive legislation still to be passed in the United States. Giostar Mexico understands that curiosty and concern is warranted; therefore, when interested patients call us to find out more about stem cell therapy in Mexico, including if it is safe and legal, we recommend everyone verifies that their chosen stem cell clinic has already received the fundamental licenses to operate a stem cell clinic in Mexico: License to Infiltrate Stem Cells and License to Bank Stem Cells. These licenses are issued by the Mexican FDA-equivalent: COFEPRIS.
Is stem cell therapy safe and legal in Mexico?
An important question our patients ask before coming to our clinics is: Is stem cell therapy safe? We also understand that, perhaps, the real question is: Is stem cell therapy legal in Mexico? Yes and of course! In Giostar Mexico, we ask for each applicant to submit a health history form, from which our medical directors personally evaluate the viability of each applicant. This means that stem cell therapy may not be for everyone. Important: we currently do not treat macular degeneration and patients with cancer.
Stem cell therapy is thoroughly regulated by the Mexican FDA-equivalent: COFEPRIS. Due to advanced legislation, Mexico enjoys sufficient regulatory framework to offer applications otherwise unavailable in countries such as the US or Canada, for example: local direct applications (hands for arthritis, shoulder pain, knee pain), lumbar and cervical applications, all which have proven better results for patients, than just receiving stem cell therapy intravenously.